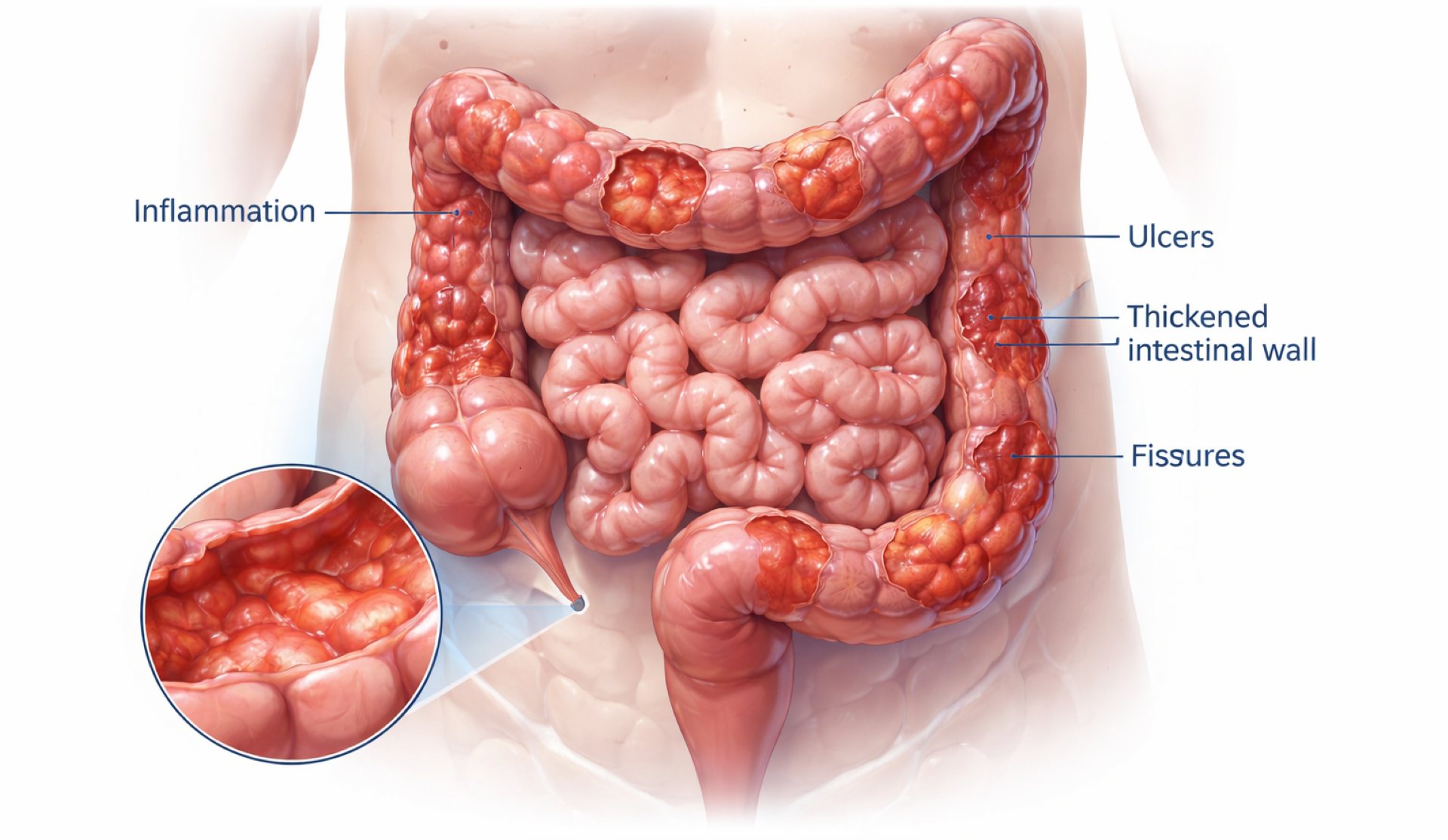
Amorphical reports positive long-term Phase 2 results for Crohn’s disease therapy
Amorphical has announced positive final long-term results from its Phase 2 clinical trial evaluating its investigational nano-amorphous Calcium Carbonate (ACC) therapy for moderate-to-severe Crohn’s disease. The data, which includes patients followed up to 48 weeks post-baseline, confirms interim findings and demonstrates sustained clinical remission when ACC is added to best available standard of care (BAT).
“The consistency of the final data and the powerful signal of sustained remission over nearly two years is a significant validation for this program and the potential of our nano-amorphous technology platform,” Eden Ben, chief executive officer of Amorphical, said. “Completing the analysis with these confirmed results de-risks our program and provides a strong foundation for our planned regulatory discussions.”
The trial was conducted at Soroka University Medical Center in Israel under ethical committee supervision. The 12-week double-blind phase enrolled 23 patients, randomized 1:1 to receive ACC or placebo, followed by an open-label extension. Patients who began the study on ACC showed more than double the reduction in Crohn’s Disease Activity Index (CDAI) scores compared with placebo, with an average decrease of 102 points versus 47 points for placebo. By week 24, mean CDAI reductions reached approximately 157 points for early ACC treatment, compared with 114 points for patients who switched from placebo, highlighting a stronger and more durable clinical improvement with early therapy.
Among patients completing at least the 48-week visit, five out of eight achieved continuous clinical remission (CDAI <150) through Week 48 and beyond. Dr Arik Segal, lead investigator from the Department of Gastroenterology at Soroka University Medical Center, said: “The sustained clinical remission observed with nano-amorphous ACC over long-term follow-up is a compelling finding for patients with moderate-to-severe Crohn’s disease. The durability of response strengthens the potential of this novel approach to deliver meaningful, lasting benefit.”
ACC is designed for localized activity in the small intestine, neutralizing acidity at inflamed sites and providing bioavailable calcium that supports epithelial repair. The therapy provides a non-immunosuppressive approach and was well tolerated throughout the trial, with no new safety concerns observed during long-term follow-up.
Amorphical is now preparing to advance ACC into the next phase of clinical development, with planned regulatory discussions and strategic funding engagements, including participation at the J.P. Morgan Healthcare Conference in San Francisco. Beyond Crohn’s disease, the company is developing additional programs for pancreatic cancer, hypoparathyroidism, and osteoporotic fractures, supported by a global patent portfolio of more than 190 patents.