Boehringer Ingelheim’s apecotrep reduces proteinuria in Phase 2 FSGS trial
Boehringer Ingelheim has reported Phase 2 clinical trial results showing its investigational asset apecotrep reduced proteinuria in people with primary focal segmental glomerulosclerosis, a rare kidney disease with high unmet medical need.
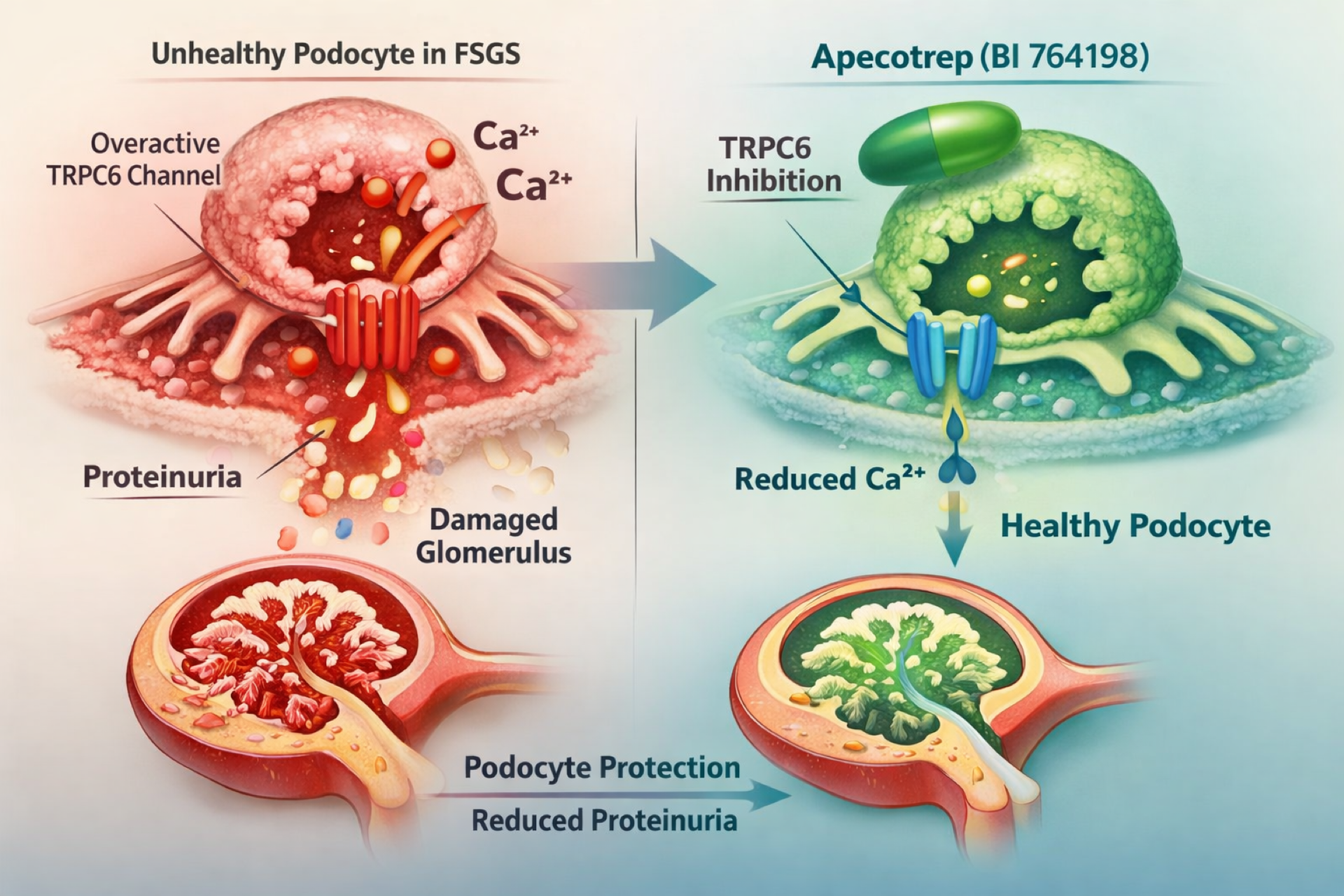
Apecotrep is an oral, selective TRPC6 inhibitor being developed as a targeted, non-immunosuppressive therapy for primary FSGS, a condition caused by injury and loss of podocytes, the cells responsible for kidney filtration. Proteinuria is a key marker of kidney damage and disease progression in FSGS.
Results from the 12-week Phase 2 study, published in The Lancet and presented at the American Society of Nephrology Kidney Week meeting, showed a 40% reduction in proteinuria in the 20 mg dose group compared with placebo. The company said the drug was generally well tolerated in adults.
A Phase 3 trial is currently recruiting adults and adolescents with primary FSGS, and an additional Phase 2 trial in other proteinuric kidney diseases is expected to start in the first quarter of this year.
Paola Casarosa, head of innovation unit, board of managing directors at Boehringer Ingelheim, said: “The results underscore Boehringer’s scientific leadership in kidney health and deep commitment to people living with cardiovascular, renal, and metabolic diseases, including rare kidney conditions like FSGS.” She added: “With the Phase 3 trial now underway, we are advancing apecotrep driven by the potential to deliver the first disease-modifying treatment for primary FSGS and redefine the standard of care for patients.”
Primary FSGS is a rare, progressive glomerular disease that can lead to kidney failure, with around half of patients progressing to end-stage kidney disease within five to ten years. There are currently no approved targeted disease-modifying therapies, leaving a significant unmet need for treatments addressing underlying disease mechanisms.
Howard Trachtman, lead investigator at the University of Michigan, said: “Primary FSGS is a serious glomerular disease and an important cause of kidney failure in both children and adults. By targeting the underlying mechanism of primary FSGS, apecotrep reduced proteinuria by 40% compared to placebo, with a favorable tolerability profile in adults.” He added that the findings support further investigation of the compound as a potential targeted therapy.
TRPC6 is thought to be overactivated in primary FSGS, leading to increased calcium influx into podocytes and progressive cellular injury. By inhibiting TRPC6, apecotrep is intended to protect podocytes and slow disease progression through reduction of proteinuria.
In the Phase 2 trial, 35% of participants receiving apecotrep across all dose groups achieved at least a 25% reduction in urine protein-creatinine ratio compared with 7.1% in the placebo group. The highest response rate was observed in the 20 mg dose cohort.
Apecotrep has received Breakthrough Therapy Designation from China’s National Medical Products Administration and Orphan Drug Designations from the European Medicines Agency and Japan’s Ministry for Health, Labour and Welfare. The compound is part of Boehringer Ingelheim’s cardiovascular, renal and metabolic portfolio.