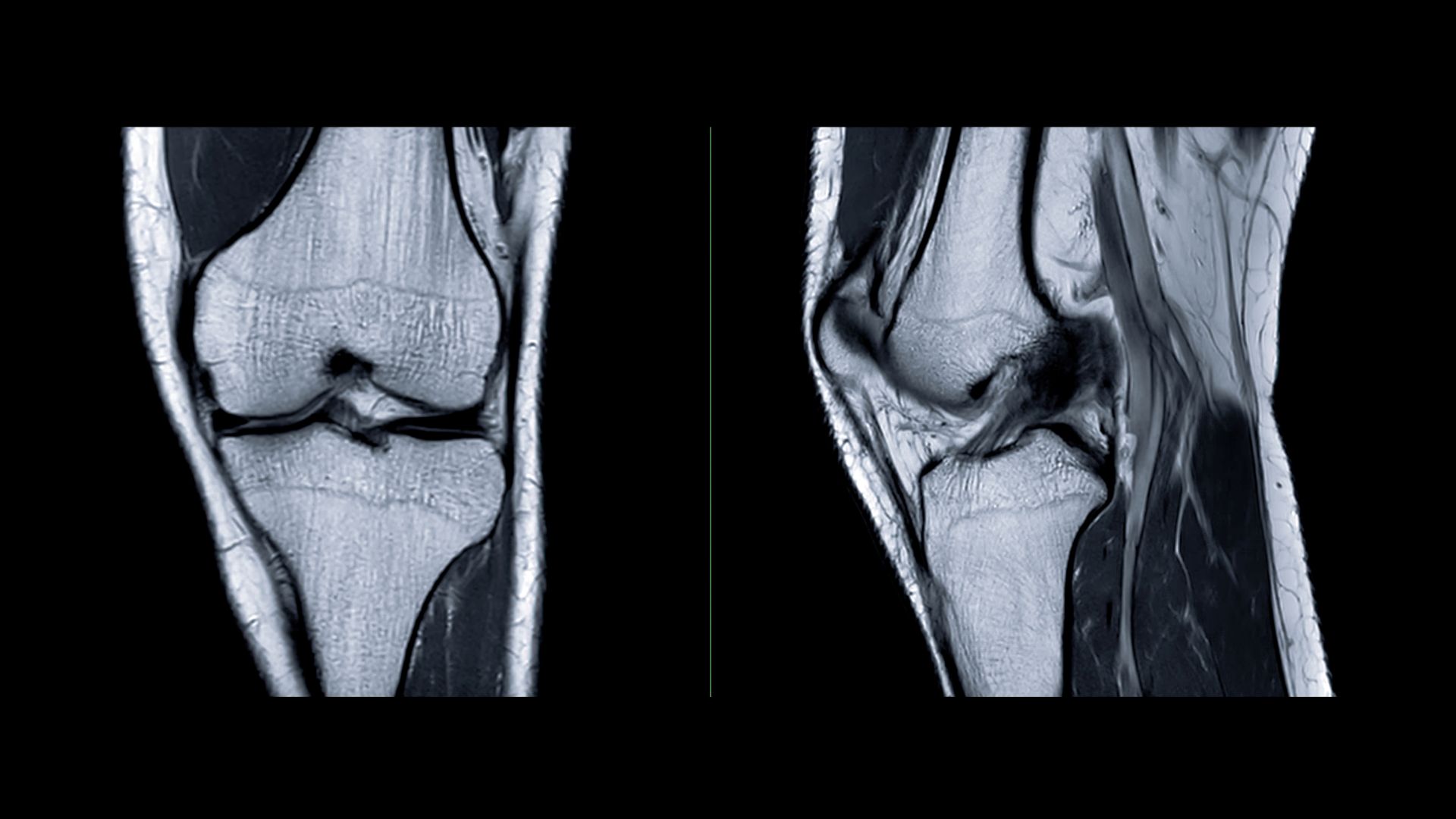
Levicept to present Phase 2 LEVI-04 data showing bone marrow lesion and pain reduction at ACR Convergence 2025
Levicept Ltd will present new findings from its Phase 2 trial of LEVI-04, a first-in-class neurotrophin-3 inhibitor, at the American College of Rheumatology’s annual meeting, ACR Convergence 2025, held from 24–29 October in Chicago. The data highlight dose-dependent reductions in bone marrow lesion presence and area in patients with osteoarthritis (OA), alongside improvements in patient-reported symptoms, including pain.
The Phase 2 study was a multiarm, multicentre, randomized, double-blind, placebo-controlled trial enrolling 518 participants with knee osteoarthritis and associated pain and disability (ClinicalTrials.gov ID: NCT05618782). Previous results from the study confirmed that LEVI-04 met its primary efficacy endpoint, demonstrating significant analgesic effects and a favourable safety and tolerability profile.
Professor Philip Conaghan MBBS, director of the NIHR Leeds Biomedical Research Centre and principal investigator, said: “We look forward to presenting new data from this large, robust study of LEVI-04 at ACR Convergence 2025. For the first time, we will show findings which suggest that LEVI-04 holds promise as a therapy to provide contemporaneous modification of joint structure—bone marrow lesions—and symptoms of OA including pain. We will also present an analysis of LEVI-04’s mechanism of action and further studies which we believe add to the evidence of LEVI-04’s potential to offer a vital new treatment option for millions of patients worldwide in need.”
Additional presentations at ACR Convergence will detail LEVI-04’s mechanism of action, functional outcomes and imaging screening exclusions, providing a comprehensive view of the biologic’s therapeutic potential. LEVI-04 is being developed as a non-addictive alternative for OA pain management, addressing both structural disease and symptomatic relief.
Levicept’s Phase 2 trial underscores the potential for LEVI-04 to combine structural joint modification with pain reduction in a patient population with limited treatment options. The results are expected to inform subsequent clinical development and regulatory strategy for the therapy.