Nanotube injector enables controlled cytoplasmic transfer between living cells
Researchers in Japan have developed a nanotube-based system that enables high-efficiency cytoplasmic transfer between living cells, overcoming a long-standing technical barrier.
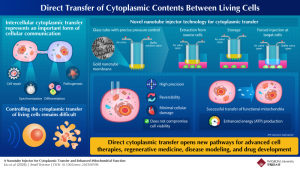
The study, led by Takeo Miyake at Waseda University and published in Small Science, addresses a long-standing limitation in cell biology: the lack of reliable tools to directly manipulate cytoplasmic contents without damaging cells. While intercellular exchange of proteins, RNA and organelles occurs naturally, existing laboratory methods to replicate or control this process have been inefficient or destructive.
Current approaches often rely on cell lysis, physical disruption or viral delivery systems. These methods can damage biomolecules, limit the size of transferable material, or prove difficult to scale. As a result, controlled cytoplasmic transfer has remained largely inaccessible despite advances in gene editing and molecular targeting.
The newly developed platform uses a thin gold membrane embedded with vertically aligned nanotubes mounted on a glass tube. When applied to cultured cells, the nanotubes penetrate the cell membrane with minimal disruption. By adjusting air pressure within the tube, researchers can extract cytoplasmic material from donor cells and subsequently deliver it into recipient cells.
Experimental results showed that transfer efficiency exceeded 90% under optimised conditions, while cell viability remained at around 95%. The team found that nanotube diameter, density and applied pressure were critical parameters in balancing efficient transfer with minimal cellular damage.
The platform was also tested for its ability to transfer functional organelles. In experiments tracking fluorescently labelled mitochondria, researchers observed that multiple mitochondria could be delivered into recipient cells. These organelles remained active, as indicated by increased adenosine triphosphate production compared to control cells.
Miyake said: “This technology establishes a new paradigm for cell manipulation—transforming cells not by genetic modification but by reconstructing intracellular composition itself.”
The findings suggest potential applications across cell therapy, disease modelling and drug screening. Direct cytoplasmic transfer could offer an alternative to genetic modification, particularly in regenerative medicine, where maintaining or restoring cellular function is a key challenge.
Miyake added: “Directly transferring healthy mitochondria or cytoplasmic components into target cells is particularly relevant for regenerative medicine, where therapeutic cells often suffer from reduced metabolic activity or functional heterogeneity after isolation and expansion. By restoring or augmenting mitochondrial function without genetic modification, the technology offers a new strategy to improve cell quality prior to transplantation.”
While the results demonstrate proof of concept, further validation will be required to assess scalability and performance in more complex biological systems. The study nonetheless highlights a potential route towards more precise manipulation of living cells, with implications for both research and clinical development.