Refold AI emerges to simplify enterprise integrations with agentic platform
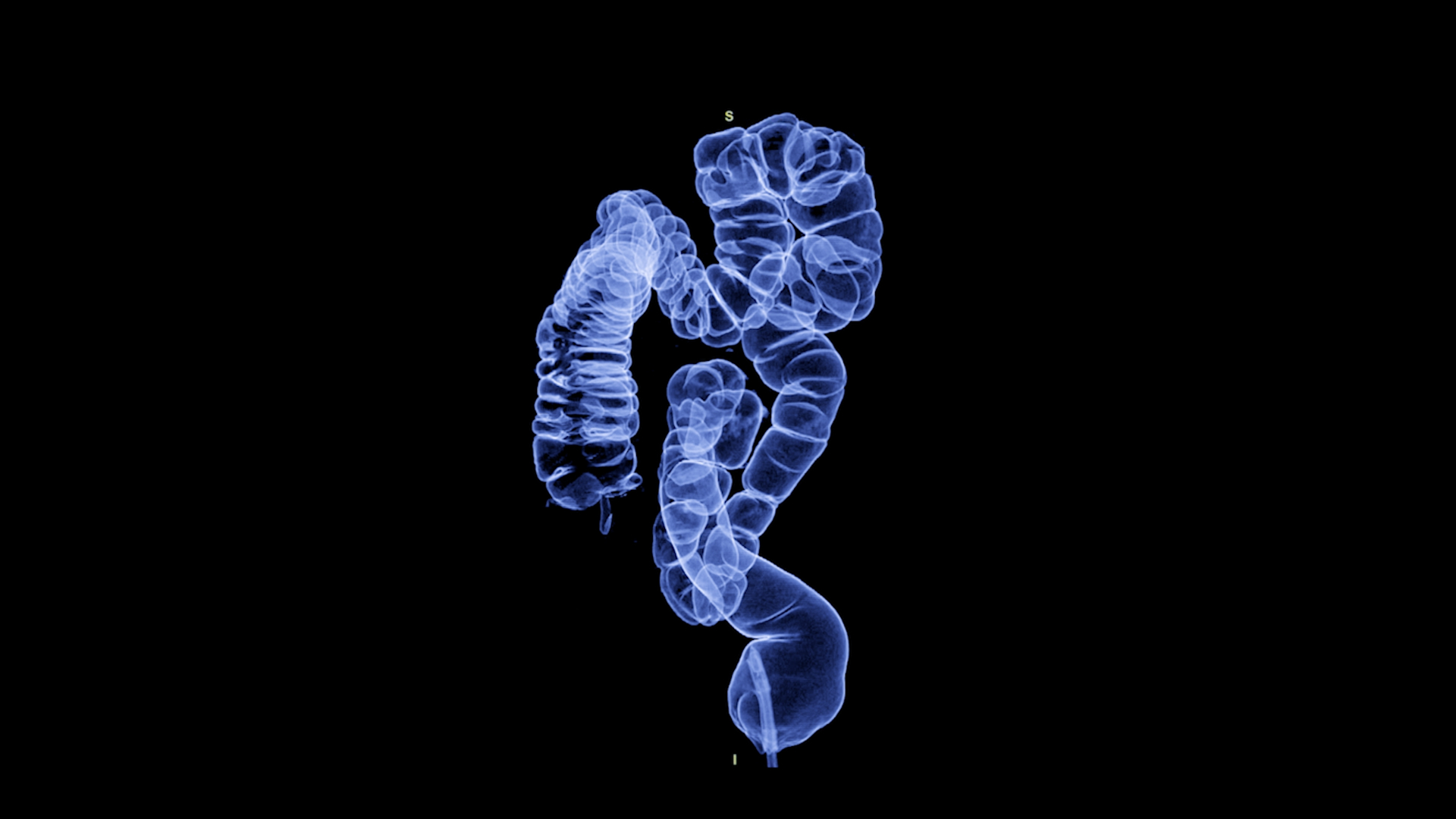
Mainz Biomed N.V., a molecular genetics diagnostic company specializing in early cancer detection, has received regulatory approval from Swissmedic to market its non-invasive colorectal cancer (CRC) screening test, ColoAlert, in Switzerland.
In Switzerland, CRC screening programs primarily target individuals aged 50 to 74, with options for a biennial fecal immunochemical test (FIT) or a colonoscopy every 10 years. This population represents roughly 2.8 million people, or around 31% of the country’s total inhabitants. Despite more than 13 screening programs nationwide, participation rates remain below 50%, leaving a significant gap in early detection coverage. Tests like ColoAlert could support national health goals by increasing participation and offering a non-invasive alternative for those reluctant to undergo colonoscopy.
The Swissmedic approval follows Mainz Biomed’s earlier partnership with a local laboratory, labor team w ag. ColoAlert will now be made available through the partner laboratory’s diagnostic services, with no remaining regulatory obstacles to its launch.
ColoAlert is designed to detect CRC tumor DNA and other biomarkers in stool samples, offering high sensitivity in a non-invasive format. It is currently marketed in select European countries, where it has been integrated into regional screening initiatives. The company aims to expand access through partnerships with laboratories and healthcare providers, ensuring localized implementation and broad availability of its molecular diagnostics.
CEO Guido Baechler said: “We are pleased to receive regulatory approval in Switzerland. This supports our broader strategy to expand access to reliable CRC screening solutions for early detection of this devastating disease and ultimately to decrease related mortality rates.”
Early detection remains a critical factor in colorectal cancer outcomes, with studies showing that timely screening can significantly reduce both incidence and mortality. By providing a non-invasive option that can be deployed through local laboratories, Mainz Biomed aims to complement existing screening programs and help reach populations that have historically had lower participation.
The company continues to focus on expanding ColoAlert’s reach across Europe, leveraging decentralized partnerships to enhance access and support national prevention efforts. Its approach reflects a broader trend in molecular diagnostics, where non-invasive tests and local lab partnerships are increasingly used to improve patient engagement, speed results, and optimize healthcare delivery.