Ultrahuman and Click Therapeutics partner on FDA-authorized digital migraine tool
Ultrahuman and Click Therapeutics have entered a partnership to develop what the companies describe as a biomarker-driven migraine management tool built on FDA-authorized digital therapeutic technology, marking a move to integrate regulated migraine interventions into a consumer wearable platform.
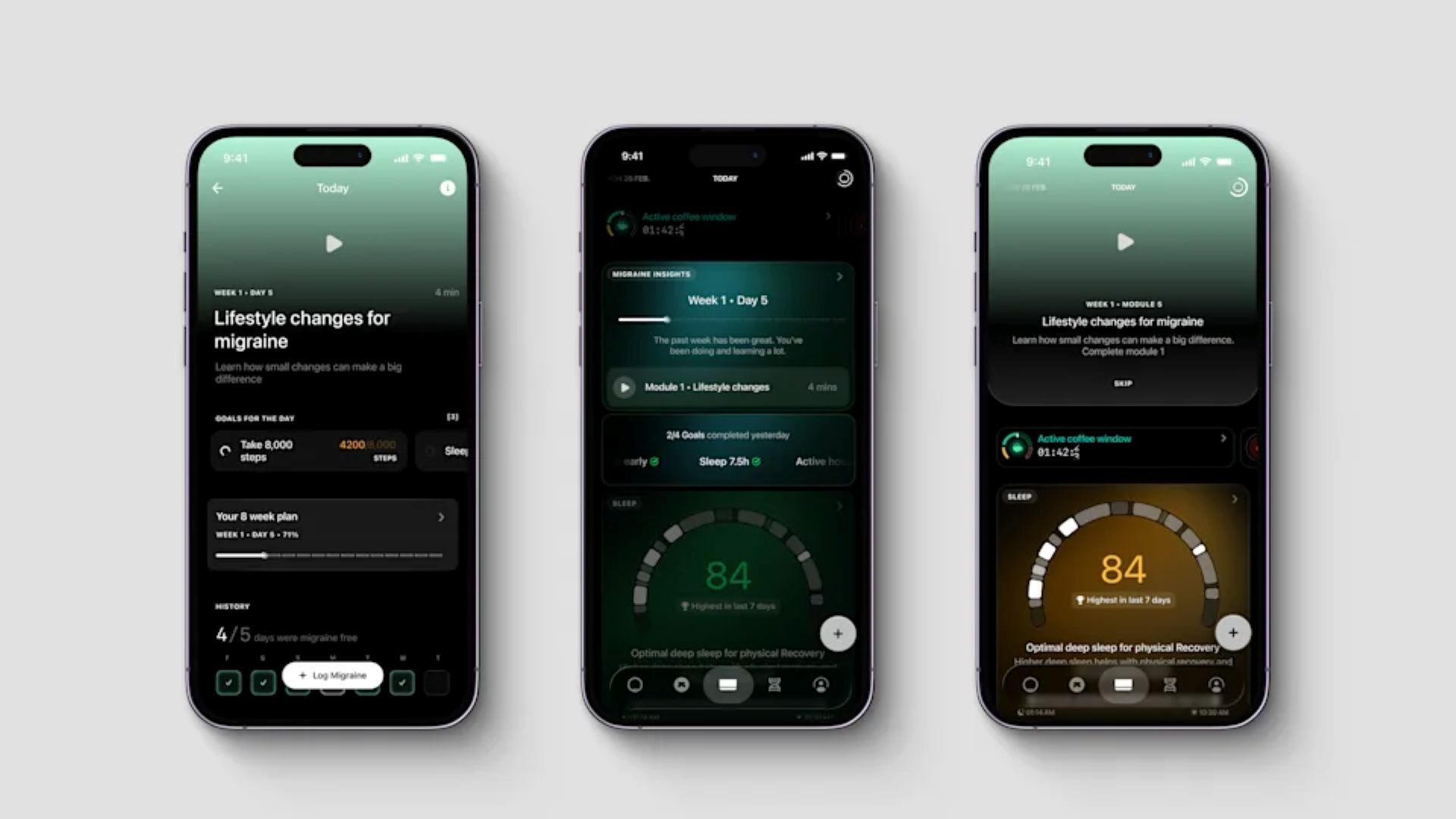
The partnership brings the technology underpinning Click Therapeutics’ FDA-authorized digital migraine treatment into Ultrahuman’s ecosystem through a new software feature called Migraine PowerPlug. The tool will sit within the Ultrahuman platform and combine wearable-derived biomarker data with guided digital interventions designed to support people living with migraine.
Migraine PowerPlug is based on the technology behind CT-132, Click Therapeutics’ FDA-authorized prescription digital therapeutic for the preventive treatment of migraine. The companies said the aim is to deliver personalized insights and guided activities informed by biometric signals such as sleep, heart rate variability, stress and movement, alongside clinically validated behavioral interventions.
Migraine affects more than one billion people globally and is estimated to impact between 15 and 20 percent of the population. The condition disproportionately affects women, who experience migraine at nearly three times the rate of men. Migraine is also cited as the leading cause of disability among women aged 15 to 49, with significant impact on work, family life and quality of life.
Ultrahuman said women’s health has been a core focus of its product roadmap, including its recent acquisition of viO and its OvuSense technology. Hormonal fluctuations are widely associated with migraine patterns, and the company believes continuous biometric data can help users better understand how these changes may relate to migraine risk, frequency and severity.
Unlike traditional migraine management, which is often limited to intermittent clinical appointments, the companies said Migraine PowerPlug is designed to support users in daily life. The platform is intended to identify correlations between physiological trends and potential migraine triggers, and then provide guided behavioral interventions informed by digital therapeutic principles rather than passive tracking alone.
Migraine PowerPlug will track trends across sleep quality, recovery, stress load and activity, translating those data into personalized lifestyle guidance. Planned features include movement and sleep goals, as well as tailored hydration recommendations based on individual migraine patterns. The companies said the goal is to help users recognize their own physiological signals and build consistency around behaviors that may improve resilience over time.
Mohit Kumar, ceo of Ultrahuman, said: “Helping people improve how they feel daily is core to Ultrahuman’s mission. By integrating the underlying principles of Click Therapeutics’ digital migraine treatment technology into our latest PowerPlug, we are enabling users to track and understand migraine-related patterns in real time, and then go further to turn those insights into actionable guidance.”
David Benshoof Klein, ceo and founder of Click Therapeutics, added: “Our partnership with Ultrahuman will allow us to bring a much-needed personalized migraine solution to people around the world. As digital health evolves, combining our clinically validated digital treatments with personalized consumer biometrics allows us to address an unmet need in over-the-counter care and the fast-growing wearables market.”
The Migraine PowerPlug is scheduled to launch in early 2026 following a pilot phase. Initial availability is planned across the United States, Canada, EU, India and Australia. The companies have not disclosed commercial terms of the partnership.